Overview
The discovery of GLP-1 and its biological activity
The sequence of GLP-1 was first identified following the cloning of the cDNAs and genes encoding proglucagon in the early 1980s, beginning with the anglerfish sequences by Lund and Goodman in the Habener lab Pancreatic pre-proglucagons are encoded by two separate mRNAs J Biol Chem. 1981 Jul 10;256(13):6515-8 and
Pancreatic preproglucagon cDNA contains two glucagon-related coding sequences arranged in tandem Proc Natl Acad Sci U S A. 1982 Jan;79(2):345-9 followed by the hamster cDNA
Hamster preproglucagon contains the sequence of glucagon and two related peptides Nature. 1983 Apr 21;302(5910):716-8 and human Exon duplication and divergence in the human preproglucagon gene Nature. 1983 Jul 28-Aug 3;304(5924):368-71 proglucagon gene in the Bell lab, and the rat cDNA Pre-proglucagon messenger ribonucleic acid: nucleotide and encoded amino acid sequences of the rat pancreatic complementary deoxyribonucleic acid Endocrinology. 1984 Dec;115(6):2176-81 and gene Glucagon gene sequence. Four of six exons encode separate functional domains of rat pre-proglucagon J Biol Chem. 1984 Nov 25;259(22):14082-7 in the Habener lab.
Following detection of the putative GLP-1 sequences within the cDNAs and genes, Dr. Svetlana Mojsov, a chemistry specialist in the Peptide Unit of the MGH, working with the Habener lab, used synthetic peptides to develop antibodies and radioimmunoassays for GLP-1 and GLP-2, enabling detection of the peptides in cells and tissues. Drucker utilized cell transfection techniques to introduce the proglucagon cDNAs into three different cell lines. endocrine (pituitary and islet cells) vs. non-endocrine cell lines (fibroblasts) transfected with the rat proglucagon cDNA. Drucker's studies whowed that endocrine cells, but not fibroblasts, demonstrated the capacity for production of multiple smaller GLP-1-immunoreactive peptides, including GLP-1(7-37) using HPLC analysis of gel filtration chromatograpy fractions Cell-specific post-translational processing of preproglucagon expressed from a metallothionein-glucagon fusion gene J Biol Chem. 1986 Jul 25;261(21):9637-43
A similar profile of GLP-1 immunoreactive peptides was detected by Mojsov in her studies of proglucagon processing using rat pancreas and intestine. Multiple immunoreactive GLP-1 peptides were detected in extracts of the rat intestine, but very similar forms of putative GLP-1 peptides were also detected in the rat pancreas. Preproglucagon gene expression in pancreas and intestine diversifies at the level of post-translational processing J Biol Chem. 1986 Sep 5;261(25):11880-9. The elution profiles of the immunoreactive peptides detected using HPLC on specific chromatography fractions were similarly consistent with detection of multiple GLP-1 peptides, including GLP-1(1-37) and GLP-1(7-37) in the rat pancreas and intestine. These two processing papers were published in JBC a few months apart in 1986.
A key next step was to ask the question-whether GLP-1 bioactive and what does it do? Drucker explored this question using the same 3 cell lines (pituitary, islet and fiborblast cells) in the lab that he had used for studies of proglucagon processing. Notably, Hoosein and Gurd had shown that GLP-1(1-36) amide stimulated adenylate cyclase in rat hypothalamic and pituitary cell membranes. Human glucagon-like peptides 1 and 2 activate rat brain adenylate cyclase FEBS Lett. 1984 Dec 3;178(1):83-6. hence it was logical to interrogate the pituitary cells; it was also well known that glucagon stimulated insulin secretion, hence it was similarly logical to interrogate the insulin-producing islet cells. Given the detection of several smaller forms of GLP-1 in the cell line processing studies, it was logical to tests multiple GLP-1 peptides to assess their bioactivity.
Drucker did not detect any activity (cyclic AMP, hormone secretion/gene expression) for the different forms of GLP-1 when the various peptides were incubated with the pituitary and fibroblast cells. In contrast, Drucker observed that only the N-terminally truncated version of GLP-1, GLP-1(7-37) but not GLP-1(1-37), increased cyclic AMP formation, insulin gene expression, and insulin secretion, notably, only when the glucose was elevated. Glucagon-like peptide I stimulates insulin gene expression and increases cyclic AMP levels in a rat islet cell line Proc Natl Acad Sci U S A. 1987 May;84(10):3434-8
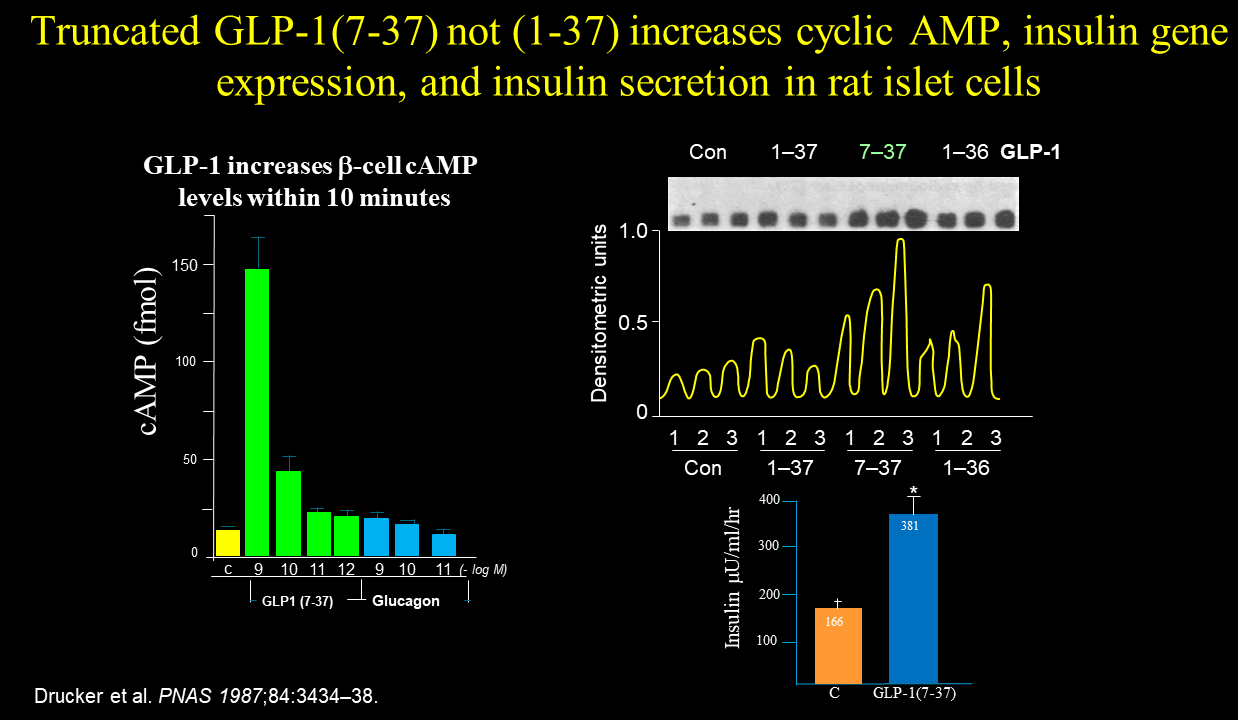
These experiments provided the first discovery of GLP-1 bioactivity in the Habener lab. Notably, there was no suggestion or direction received from others instructing Drucker to carry out the studies interrogating GLP-1 bioactivity using the cell lines he had in the lab.
Consistent with this chronology and narrative, Mojsov et al in the Discussion section of the 1986 JBC paper analyzing the processing of proglucagon disclosed that the Habener lab had detected an insulinotropic activity of GLP-1 on islet cells in vitro, referencing, as a footnote, Drucker, D.J. et al, manuscript in preparation

After the Drucker studies describing the direct insulinotropic actions of GLP-1(7-37) using insulin-producing islet cells, Habener and Mojsov approached Gordon Weir working at the Joslin, who had successfully developed a perfused rat pancreas system in his laboratory, suitable for studying the insulinotropic actions of different secretagogues. Dr. Weir tested several of the GLP-1 peptides provided by Dr. Mojsov and the Habener lab, and confirmed that GLP-1(7-37) was a potent insulinotropic peptide in the perfused rat pancreas, consistent with the first demonstration of insulinotropic activity in experiments carried out by Drucker in the islet cell studies in the Habener lab. In contrast to the islet studies carried out by Drucker et al, that showed a clear glucose-dependent potentiation of GLP-1's actions to stimulate insulin secretion, the perfused rat pancreas studies examined GLP-1 action at only a single glucose concentration (~119 mg% or 6.6 mM glucose). The Mojsov et al paper describing the perfused pancreas studies done by Gordon Weir in collaboration with the Habener group and Dr. Mojsov were published several months before the Drucker et al PNAS paper Insulinotropin: glucagon-like peptide I (7-37) co-encoded in the glucagon gene is a potent stimulator of insulin release in the perfused rat pancreas J Clin Inv 1987 Feb;79(2):616-9
Simultaneously, studies in Copenhagen led by Jens Holst and colleagues also demonstrated than a smaller N-terminally truncated form of GLP-1, GLP-1(7-36) amide purified from the gut, was a potent stimulator of insulin secretion in the perfused pig pancreas model. Notably, these studies also used only a single glucose concentration (7 mM), and hence did not examine the glucose-dependent potentiaion of insulin secretion by GLP-1 Truncated glucagon-like peptide I, an insulin-releasing hormone from the distal gut FEBS Lett. 1987 Jan 26;211(2):169-74 In related studies, the Holst lab also examined the postranslational processing of proglucagon in pancreas vs. gut, demonstrating liberation of smaller GLP-1 peptides in the human gut Pancreatic and intestinal processing of proglucagon in man Diabetologia Nov;30(11):874-81
Intriguingly, Joel Habener filed multiple patents using the totality of the Drucker and Mojsov data, as the sole inventor. Although Mojsov successfully contested and sued to revise the inventorship status of several patents, Drucker declined independent lawyers offers/advice to also sue Dr. Habener and the Massachusetts General Hospital in regard to contested contributions and inventorship.
GLP-1, food intake, weight loss and the treatment of obesity
The first observations that GLP-1 inhibits food intake came about 10 years later, in 1996. Turton and Bloom demonstrated that icv GLP-1 inhibited food intake and rats, actions blocked by icv administration of the GLP-1 receptor antagonist, exendin(9-39). A role for glucagon-like peptide-1 in the central regulation of feeding Nature 1996 Jan 4;379(6560):69-72. These findings were replicated several months later by Tang-Christensen and colleagues as well as by Scrocchi et al in the Drucker lab. The mouse studies in Toronto demonstrated that icv GLP-1 reduced food intake and attenuated the induction of food intake by NPY, actions that were absent in Glp1r-/- mice Glucose intolerance but normal satiety in mice with a null mutation in the glucagon-like peptide 1 receptor gene Nature Medicine 1996 Nov;2(11):1254-8.
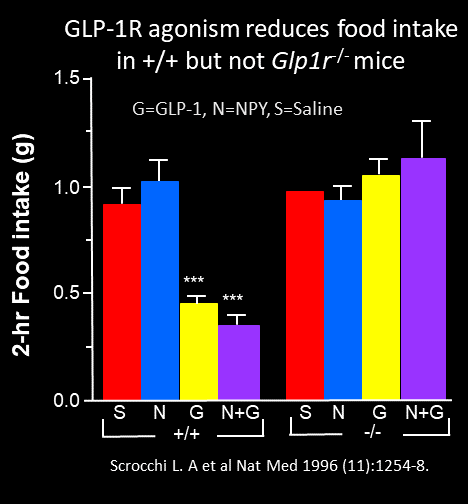
Notably, the Drucker lab had also originally sent their paper to Nature, but after much back and forth, the Editors were disappointed tha the Glp1r-/- mouse did not eat more and become obese. Hence publication of the paper was delayed, and it ultimately ended up in Nature Medicine. These preclinical findings were widely replicated and years later, it was observed that with increasing doses of GLP-1R agonists, substantial weight loss could be achieved in humans, leading to the first approval of liraglutide for the treatment of obesity in 2014.
The acute insulinotropic actions of GLP-1(7-36) in health non-diabetic humans, studied both in the fasted state, and with intravenous glucose administration, was first demonstrated by Kreymann, Bloom and colleagues later in 1987 Glucagon-like peptide-1 7-36: a physiological incretin in man Lancet. 1987 Dec 5;2(8571):1300-4.
----------------------------------------------------------------------------------------------------------------------------------------------------------
Glucagon-like peptide-1 was subsequently shown to be synthesized in intestinal endocrine cells in 2 principal major molecular forms, as GLP-1(7-36)amide and GLP-1(7-37). As noted above, the putative peptide sequence was first identified following the cloning of cDNAs and genes for proglucagon in the early 1980s, and is now known to be one of the two principal incretin hormones.
The majority of circulating biologically active GLP-1 is found in the GLP-1(7-36)amide form), with lesser amounts of the bioactive GLP-1(7-37) form also detectable. See Tissue and plasma concentrations of amidated and glycine-extended glucagon-like peptide I in humans. Diabetes. 1994 Apr;43(4):535-9 for the human data. Both peptides appear equipotent in all biological paradigms studied to date Biological effects and metabolic rates of glucagonlike peptide-1 7-36 amide and glucagonlike peptide-1 7-37 in healthy subjects are indistinguishable. Diabetes. 1993 May;42(5):658-61. GLP-1 is secreted from gut endocrine cells in response to nutrient ingestion and plays multiple roles in metabolic homeostasis following nutrient absorption.
An important locus for regulation of GLP-1 biological activity is the N-terminal degradation of the peptide by Dipeptidyl Peptidase-4 (DPP-4)-mediated cleavage at the position 2 alanine. For an overview, see DPP-4.
The biological activities of GLP-1 include stimulation of glucose-dependent insulin secretion and insulin biosynthesis, inhibition of glucagon secretion and gastric emptying, and inhibition of food intake. GLP-1 appears to have a number of additional effects in the GI tract and central nervous system, as reviewed in Diabetes 1998 47(2):159-69, Minireview: the glucagon-like peptides. Endocrinology. 2001 Feb;142(2):521-7, Development of glucagon-like peptide-1-based pharmaceuticals as therapeutic agents for the treatment of diabetes. Curr Pharm Des. 2001 Sep;7(14):1399-412. Review and Biological actions and therapeutic potential of the glucagon-like peptides. Gastroenterology. 2002 Feb;122(2):531-44, Pharmacology, physiology, and mechanisms of incretin hormone action Cell Metab. 2013 Jun 4;17(6):819-37 and Deciphering metabolic messages from the gut drives therapeutic innovation: the 2014 Banting Lecture Diabetes. 2015 Feb;64(2):317-26.
The finding that GLP-1 lowers blood glucose in patients with diabetes, taken together with suggestions that GLP-1 may restore β-cell sensitivity to exogenous secretagogues, suggests that augmenting GLP-1 signaling is a useful strategy for treatment of diabetic patients. There are a number of different GLP-1 targets or loci that may be exploited to enhance GLP-1 action in diabetic subjects.
Mounting evidence demonstrates that GLP-1R signaling regulates islet proliferation and islet neogenesis. For a review of the data to date, see GLP-1 and islet proliferation.
GLP-1 and the CNS. There is considerable interest in understanding the role (s) of GLP-1 in the control of satiety and food intake. A large body of evidence demonstrates that ICV GLP-1 can reduce food intake in both acute and chronic studies. Conversely, ICV administration of the GLP-1 antagonist exendin (9-39) can acutely increase food intake and promote weight gain in chronic rodent studies. These satiety-related effects have also been observed in human studies with peripheral administration of GLP-1 or Exenatide or Liraglutide to both normal and diabetic subjects. Nevertheless, there remains some controversy as to whether the satiety-related effects of GLP-1 are specific, or perhaps indicative of GLP-1 effects on brain regulatory networks that respond to stress or aversive conditions. For an overview of different aspects of GLP-1 biology, see:
GLP-1, β-cell proliferation and differentiation and apoptosis
GLP-1 and islet transplantation
GLP-1 and the cardiovascular system
GLP-1 actions independent of glucose homeostasis
GLP-1 Receptor Desensitization
GLP-1 actions and a second GLP-1 receptor
GLP-1 synthesis and secretion, and degradation
GLP-1 action in human subjects
Alternative GLP-1 delivery systems
