For more information on Vildagliptin, see Vildagliptin/Galvus
Sustained Reduction in HbA1c during One-Year Treatment with Vildagliptin in Patients with Type 2 Diabetes (T2DM)
Year: 2006 Abstract Number: 120-OR Authors: SYLVIE DEJAGER, ALEXANDRE LEBEAUT, ANDRE COUTURIER, ANJA SCHWEIZE R. Institutions: E Hanover, NJ; Basel, Switzerland.
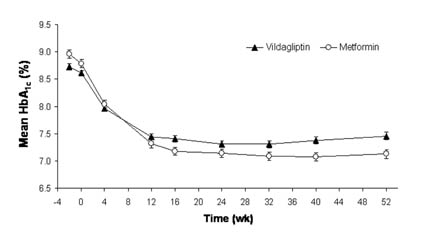
Results: Vildagliptin is a potent and selective DPP-4 inhibitor that improves islet function by increasing ?- and ?-cell responsiveness to glucose. This 52-wk, multicenter, randomized, double-blind study compared vildagliptin 100 mg daily (50 mg bid, n=526) to metformin 2000 mg daily (Met, 1000 mg bid, n=254) in drug-naive patients (pts) with T2DM. The groups were well-balanced at baseline (BL). Mean age, BMI, HbA1c and disease duration were 53.1 y, 32.4 kg/m?2, 8.7% and 2.4 y, respectively. As shown below, both vildagliptin and Met rapidly decreased HbA?1c and this improvement was sustained throughout 52-wk treatment. The adjusted mean change (AM?) from BL to endpoint was -1.0±0.1% in pts receiving vildagliptin and -1.4±0.1% in those receiving Met. The between-group difference in AM? HbA?1c did not establish non-inferiority of vildagliptin 100 mg daily to Met 2000 mg daily in these drug-naive pts. [figure1] Vildagliptin did not induce weight gain (0.3±0.2 kg) and Met-treated pts had a modest weight loss (-1.9±0.3 kg). The incidence of side effects was 70.1% in patients receiving vildagliptin vs 75.4% with Met. The incidence of GI side effects was significantly less in pts receiving vildagliptin (21.8%) vs Met (43.7%, P<0.001). Diarrhea was reported by 6.0 vs 26.2% of pts receiving vildagliptin vs Met; nausea, by 3.3 vs 10.3%; abdominal pain, by 2.3 vs 7.1%; dyspepsia, by 1.2 vs 4.8%; and vomiting, by 2.1 vs 4.4%. A mild hypoglycemic episode was reported by 3 vildagliptin-treated patients (0.6%) and by one Met-treated patient (0.4%). Conclusions: 1) a clinically meaningful reduction in HbA?1c is sustained throughout one-year treatment with vildagliptin 50 mg bid, 2) vildagliptin is well-tolerated and associated with fewer GI side effects than metformin.
 Vildagliptin Added to Metformin Improves Glycemic Control and May Mitigate Metformin-Induced GI Side Effects in Patients with Type 2 Diabetes (T2DM)
Vildagliptin Added to Metformin Improves Glycemic Control and May Mitigate Metformin-Induced GI Side Effects in Patients with Type 2 Diabetes (T2DM)
Year: 2006 Abstract Number: 121-OR Authors: ALAN GARBER, RICCARDO P. CAMISASCA, ELENA EHRSAM, CAROLE COLLOBER-MAUGEAIS, ERIKA ROCHOTTE, ALEXANDRE LEBEAUT. Institutions: Houston, TX; Basel, Switzerland; E Hanover, NJ.
Results: Vildagliptin is a potent and selective DPP-4 inhibitor that improves islet function by increasing ?- and ?-cell responsiveness to glucose. We report findings of a 24-wk, multicenter, double-blind, randomized, parallel group study comparing vildagliptin (50 mg qd or bid) to placebo (PBO) in 416 patients (pts) with T2DM continuing a previous stable metformin regimen (=1500 mg/d). The 3 treatment groups were well-balanced at baseline (BL). Mean age=56 y, BMI= 32.8 kg/m?2, disease duration=6.2 y, HbA?1c= 8.4%, metformin dose=2102 mg/d and duration of use=17.2 mo. Figure 1 depicts mean HbA1c and FPG during the 24-wk study. The between-group difference in the adjusted mean change (AM?) in HbA1c was -0.7±0.1% (P<0.001 vs PBO) in pts receiving vildagliptin 50 mg qd and -1.1±0.1% (P<0.001 vs PBO) in pts receiving 50 mg bid. The between-group difference in the AM? in FPG was -0.8 ±0.3 mM (P=0.003 vs PBO) and -1.7±0.3 mM (P<0.001 vs PBO) in pts receiving vildagliptin 50 mg qd and 50 mg bid, respectively. [figure1]There was no weight gain in the 2 vildagliptin groups and the incidence of side effects did not differ between groups (63.3, 65.0 and 63.5% of pts randomized to vildagliptin 50 mg qd, 50 mg bid and PBO, respectively). Of note, 9.6%, 14.8% and 18.2% of pts receiving vildagliptin 50 mg qd, 50 mg bid and PBO reported =1 GI side effect and the incidence in the vildagliptin 50 mg qd group was significantly lower (P=0.022) than in the PBO group. One hypoglycemic event occurred in each group. Conclusions: vildagliptin is well-tolerated and produces clinically-meaningful, dose-related decreases in HbA?1c and FPG in metformin-treated pts with T2DM.
Vildagliptin Suppresses Endogenous Glucose Production (EGP) and Increases Beta Cell Function after Single Dose Administration in Type 2 Diabetic (T2D) Patients
Presented during: Year: 2006 Abstract Number: 122-OR Authors: BOGDAN BALAS, MUHAMMAD BAIG, CATHERINE WATSON, BETH E. DUNNING, MONICA LIGUEROS-SAYLAN, YAN-LING HE, YIBIN WANG, KENNETH CUSI, JAMES E. FOLEY, RALPH A. DEFRONZO.
Institutions: San Antonio, TX; East Hanover, NJ. Results:
Introduction: Vildagliptin is a selective DPP-4 inhibitor that augments GLP-1 levels. Chronic vildagliptin treatment decreases postprandial glucose levels and reduces HbA?1c in T2D. However, little is known about the mechanisms by which vildagliptin promotes reduction in fasting plasma glucose (FPG) conc.
Aim: To examine the mechanisms via which vildagliptin improves glucose tolerance and reduces FPG in T2D.
Methods: 16 T2D (age=48±12y; BMI=34.4±6.7; HbA?1c=9±0.3%) participated in a randomized, double-blind, placebo (PBO)-controlled, trial. On separate days patients received 100 mg vildagliptin or PBO at 5:30PM followed 30 min later by 6-h meal tolerance test (MTT) performed with double tracer technique (3-?3H-GLU iv and 1-?14C-GLU incorporated in meal) to assess hepatic and peripheral glucose metabolism. Following vildagliptin, suppression of EGP during 6-h MTT was greater than with PBO (1.02±0.06 vs 0.74±0.06 mg/kg•min, p=0.004) while rate of total glucose disposal (TGD) was similar. During 6-h MTT performed with vildagliptin, insulin secretion rate (ISR) increased by 21% (p=0.003 vs PBO) despite significant reduction in mean plasma glucose (213±4 vs 230±4 mg/dl, p=0.006); consequently, the insulinogeneic index (?ISR÷?plasma glucose) increased by 29% (p=0.01). Suppression of mean plasma glucagon during MTT was 93% greater with vildagliptin (p<0.01). From 6PM to 8AM EGP was significantly reduced (0.8±0.06 vs 0.5±0.06 mg/kg•min, p<0.003) in vildagliptin vs PBO and the decline in EGP was positively correlated (r=0.48, p<0.006) with the 14mg/dl decrease in FPG observed at 8AM.
Conclusions: During MTT, vildagliptin augments insulin secretion and inhibits glucagon release, leading to enhanced suppression of EGP. During the entire overnight period (6PM-8AM), a single dose of vildagliptin reduced FPG by enhancing suppression of EGP.
Restoration of an Acute Insulin Response to Glucose (AIRg) in Drug-Naive Patients with Type Diabetes (T2DM) by 3-Month Treatment with Vildagliptin
Year: 2006 Abstract Number: 454-P Authors:
DAVID A. D'ALESSIO, CATHERINE E. WATSON, YAN-LING HE, MONICA LIGUEROS-SAYLAN, YIBIN WANG, BETH E. DUNNING, RONALD L. PRIGEON, JAMES E. FOLEY, RICHARD E. PRATLEY. Institutions: Cincinnati, OH; Cambridge, MA; E Hanover, NJ; Princeton, NJ; Baltimore, MD; Burlington, VT.
Results: Development of T2DM is characterized by loss of the AIRg and impaired insulin sensitivity (Si). This study examined the effect of the DPP-4 inhibitor vildagliptin (50 mg bid, n=7) or placebo (PBO, n=5) on AIRg and Si determined during frequently-sampled IV glucose tolerance tests (FSIVGT) performed in drug-naive patients (pts) with T2DM before and after 12-wk treatment (Rx) and after 2-4 wk washout from study drug. At baseline (BL) mean HbA?1c, BMI, age and disease duration were 6.8±0.1%, 30.9±1.8 kg/m?2, 56±2.5 y and 3.9±0.8 y. Plasma levels of intact GLP-1 were not significantly increased during the FSIVGTs which were performed after overnight fast. In the PBO group, AIRg and Si did not change from BL to Wk 12. In vildagliptin-treated pts, AIRg increased from 16±7 at BL to 54±9 pM at Wk 12 (P<0.001 vs BL or PBO); Si increased from 1.4±0.3 at BL to 2.2±0.5 L/mU/min at Wk 12 (P=0.016 vs BL). Figure 1 depicts the disposition index (DI=AIRg x Si) at BL, Wk 12 and after washout. The DI increased >4-fold during vildagliptin Rx and a part of this effect remained after washout.[figure1]HbA?1c decreased from 6.7±0.5% at BL to 6.3±0.5% at Wk 12 (?=-0.5±0.3, P=0.013) in the vildagliptin group but did not change from BL (6.9%) in the PBO group. Conclusion: 12-wk Rx with vildagliptin improves ?-cell function and attenuates insulin resistance in drug-naive pts with T2DM independent of acute increases in plasma GLP-1. Because a part of the improvement is maintained after a 2-4 wk washout, vildagliptin may exert some disease-modifying effect.
Vildagliptin as Add-On to Insulin in Patients with Type 2 Diabetes (T2DM)
Year: 2006 Abstract Number: 467-P Authors: VIVIAN FONSECA, SYLVIE DEJAGER, DIEGO ALBRECHT, LYNDA SHIRT, ANJA SCHWEIZE R. Institutions: New Orleans, LA; Basel, Switzerland; E Hanover, NJ.
Results: Vildagliptin is a DPP-4 inhibitor that lowers glucose in patients (pts) with T2DM primarily via glucagon-like peptide-1 (GLP-1)-mediated improvements in islet function. Vildagliptin decreases HbA1c when given as monotherapy or in combination with metformin but its efficacy in pts with more advanced disease requiring insulin (Ins) is unknown. This 24-wk, multicenter, double-blind, randomized study compared effects of vildagliptin 50 mg bid vs placebo (PBO) in 256 pts with T2DM inadequately controlled (HbA?1c 7.5-11%) by Ins (>30 U/day). Pts had received no oral agent in the previous 3 mos and Ins dose adjustments were allowed during the study. The treatment groups were well-balanced at baseline (BL). Mean age, BMI, disease duration, FPG and HbA?1c were 59 y (32% over age 65), 33.0 kg/m?2, 14.6 y, 9.3 mM and 8.5%, respectively. Mean duration of Ins use was 76 mo, mean daily dose was 82 U. Pts had multiple co-morbidities and 45% were taking >5 concomitant medications. Table 1 reports the adjusted mean change (AM?) from BL to endpoint in HbA1c and daily Ins dose in the two treatment groups. [table1] In pts =65 y, HbA1c decreased by 0.7±0.1% in the vildagliptin group vs 0.0±0.1% in the PBO group (P<0.001). Hypoglycemic events were less common and less severe in vildagliptin-treated pts (33 patients, 113 events, 0 severe) than in those receiving PBO (45 patients, 185 events, 6 severe). The incidence of side effects was similar in pts receiving vildagliptin vs PBO. Conclusion: adding vildagliptin to Ins in pts with T2DM is well-tolerated and associated with a significant reduction in HbA1c, particularly in older individuals, and despite clinical insulin resistance. The mechanism underlying the reduced frequency and severity of hypoglycemia in vildagliptin-treated pts receiving Ins may reflect improved ?-cell glucose sensing and merits further investigation.
Reduction in Blood Pressure in Patients Treated with Vildagliptin as Monotherapy or in Combination with Metformin for Type 2 Diabetes
Year: 2006 Abstract Number: 474-P Authors: AMEET NATHWANI, ALEXANDRE LEBEAUT, STUART BYIERS, CLAUDIO GIMPELEWICZ, IH CHANG. Institutions: Basel, Switzerland; East Hanover, NJ.
Results: Vildagliptin is a potent and selective DPP-4 inhibitor that improves islet function by increasing ?- and ?-cell responsiveness to glucose. Vildagliptin decreases HbA?1c when given as monotherapy or in combination with metformin. Because hypertension is an important cardiovascular risk factor in diabetic patients, the effect of vildagliptin on blood pressure was evaluated. In a 24-week multicenter, randomized, double-blind placebo controlled study of vildagliptin given as monotherapy in treatment-naive patients with type 2 diabetes, mean reductions of 1.4/1.0 mmHg, 4.1/2.7 mmHg and 3.0/1.4 mmHg in sitting systolic/diastolic blood pressure (measured at the clinic) at the end of 24 week treatment from mean baselines of 131.5/80.6 mmHg, 133.0/81.4 mmHg and 131.7/80.9 mmHg were seen in patients treated with vildagliptin 50 mg qd (n=155), 50 mg bid (n=145) and 100 mg qd (n=154) respectively, as compared to a mean reduction of 1.5/0.9 mmHg from baseline of 131.3/81.2 mmHg in the placebo treated patients (n=151). In a 24-week multicenter, randomized, double-blind placebo controlled study comparing vildagliptin (50 mg qd or bid) to placebo in patients with type 2 diabetes continuing a previous stable metformin regimen (=1500 mg/d), mean reductions of 2.0/0.8 mmHg and 3.5/2.2 mmHg in sitting systolic/diastolic blood pressure (measured at the clinic) at the end of 24 week treatment from mean baselines of 131.2/80.2 mmHg and 134.1/80.8 mmHg were seen in patients treated with vildagliptin 50 mg qd (n=174) and 50 mg bid (n=176) respectively, as compared to a mean reduction of 0.8/0.1 mmHg from baseline of 133.3/79.8 mmHg in the placebo group (n=175). These reductions suggest a modest positive effect of vildagliptin on blood pressure in patients with type 2 diabetes.
The Insulinotropic Effect of Vildagliptin Is Revealed When Oral Glucose Tolerance Tests (OGTT) Impose Large Glucose Loads
Year: 2006 Abstract Number: 483-P Authors: YAN-LING HE, JENS J. HOLST, CAROLYN F. DEACON, BETH E. DUNNING, MONICA LIGUEROS-SAYLAN, JAMES E. FOLEY.
Institutions: Cambridge, MA; Copenhagen, Denmark; Princeton, NJ; E Hanover, NJ.
Results: Although vildagliptin improves ?-cell function and reduces postprandial glucose (PPG) in patients (pts) with type 2 diabetes (T2DM), it does not increase post-meal insulin levels during 4-12 wk treatment in pts with T2DM, nor after a single dose given before a 75-g OGTT in healthy subjects. To explore this apparent conundrum, we extract data from two studies of vildagliptin in diet-treated pts with T2DM. In Study 1, 16 pts (mean BMI, HbA?1c, FPG and fasting insulin = 30.0 kg/m?2, 8.0%, 8.3 mM and 96 pM) received a single oral dose of vildagliptin (100 mg) or placebo (PBO) in a randomized crossover manner with =72 h interdose interval, 30 min before a 75-g OGTT. In Study 2, 9 pts (mean BMI, HbA?1c, FPG and fasting insulin = 31.8 kg/m?2, 7.5%, 8.9 mM and 212 pM) received PBO on Day -1 and vildagliptin (100 mg) on Day 1, 30 min before a breakfast meal test, during a 28-day, double-blind, randomized, PBO-controlled trial. Table 1 summarizes DPP-4 activity at 2-h post-dose, mean post-load plasma levels of intact GLP-1, GIP and insulin (AUC/time), and plasma glucose levels 1.5 h post-load. A single dose of vildagliptin produced essentially complete inhibition of DPP-4 and increased post-load levels of intact GLP-1 and GIP during OGTT or meal test. The mixed meal was a stronger stimulus than OGTT for GIP release, but the glucose load imposed by OGTT, as reflected by peak glucose levels, was nearly twice that of the mixed meal. Vildagliptin significantly increased plasma insulin levels after OGTT but not after the mixed meal.[table1]Conclusion: an acute insulinotropic effect of vildagliptin is observed in patients with T2DM only when the glucose load provides a strong independent stimulus for insulin secretion
Vildagliptin Decreases Postprandial Chylomicron Triglycerides (TGs) in Drug-Naive Patients with Type 2 Diabetes (T2DM)
Year: 2006 Abstract Number: 524-P Authors:
NIINA MATIKAINEN, SAKARI MANTTARI, ANJA SCHWEIZER, ANNE ULVESTAD, DAVID MILLS, BETH E. DUNNING, JAMES E. FOLEY, MARJA-RIITTA TASKINEN.
Institutions: Helsinki, Finland; Basel, Switzerland; Princeton, NJ; E Hanover, NJ.
Results: Vildagliptin is a DPP-4 inhibitor that improves glycemic control via incretin hormone-mediated improvements in islet function. Data from animal studies suggest that GLP-1 can reduce intestinal TG absorption and GIP improves chylomicron TG clearance. The present 4-wk study examined effects of vildagliptin 50 mg bid (n=15) vs placebo (PBO, n=16) on postprandial lipemia in drug-naive patients (pts) with T2DM. A lipid-rich meal was given before and after 4-wk Rx: total serum TGs and cholesterol, and TGs, cholesterol and apoB-48 in the chylomicron (Sf>400) and VLDL/IDL (Sf 12-400) fractions were measured for 8-h post-meal. In pts randomized to PBO, postprandial lipids did not change or tended to increase from Wk 0 to Wk 4. In contrast, as shown below, 4-wk Rx with vildagliptin markedly decreased post-meal total serum and chylomicron TGs. [figure1] Relative to PBO, 4-wk Rx with vildagliptin decreased the AUC0-8h for total TG by 22±11% (P=0.037), the incremental (?) AUC?0-8h for total TG by 85±47% (P=0.065), the AUC?0-8h for chylomicron TGs by 65±19% (P=0.001) and the ?AUC?0-8h for chylomicron TGs by 91±28% (P=0.002). This was associated with a decrease in chylomicron apoB-48 AUC?0-8h (?= -1.0±0.5mg/L•h, P=0.037) and chylomicron cholesterol AUC 0-8h (?= -0.14±0.07 mM•h, P=0.046). Consistent with previous studies, relative to PBO, 4-wk Rx with vildagliptin also increased intact GLP-1 and decreased glucagon, fasting and postprandial glucose and HbA?1c (from a baseline of 6.7%, ?= -0.4±0.1%, P<0.001). Conclusion: vildagliptin decreases postprandial lipemia by decreasing chylomicron production and/or increasing chylomicron clearance.
Vildagliptin Improves Glucose Control as Evidenced by HbA1c after 12 Weeks Therapy in Japanese Patients with Type 2 Diabetes
Year: 2006 Abstract Number: 527-P Authors: NOBOYUKI MIMORI, SHINJI TERAO, DAVID HOLMES. Institutions: Tokyo, Japan.
Results: Vildagliptin is a potent and selective DPP-4 inhibitor that improves islet function by increasing ?- and ?-cell responsiveness to glucose. This randomized, double-blind study assessed the efficacy and tolerability of vildagliptin vs. placebo (PBO) in Japanese drug-naive patients with type 2 diabetes (T2DM). A total of 219 patients (mean ±SD age 59±8.4 years, mean BMI 24.4±2.8 kg/m?2 were randomized to PBO (N=72, mean HbA?1c 7.4±0.75%, mean fasting plasma glucose (FPG) 9.0±1.7 mmol/L) or to vildagliptin at a dosage of 10 mg bid (N=71, mean HbA?1c 7.4±0.81%, mean FPG 8.9±1.9 mmol/L), 25 mg bid (N=72, mean HbA?1c 7.4±0.9%, mean FPG 9.0±1.8 mmol/L) or 50 mg bid (N=76, mean HbA?1c 7.4±0.8%, mean FPG 8.9±1.8 mmol/L) given for 12 weeks. A standard meal test (SMT, Japanese meal, kcal 470, CHO 73%, protein 12%, fat 15%) was conducted at baseline and week 12 in a subset of patients. After 12 weeks, HbA?1c (mean±SE) increased by 0.28±0.07% ( PBO) and decreased by 0.53±0.07%, 0.67±0.07% and 0.92±0.07% (vildagliptin 10, 25 and 50 mg bid respectively, p<0.001 for all vs. PBO). FPG increased by 0.13±0.19 mmol/L (PBO) and decreased by 62±0.19, 0.78±0.19 and 1.37±0.19 mmol/L (vildagliptin 10, 25 and 50 mg bid respectively, p<0.001 for all vs. PBO). Following the SMT, peak prandial GLP-1(7-36) increased about 2-fold at week 12. The 2-hr prandial glucose level increased by 0.2 mmol/L (PBO) and was reduced by 3.5, 3.2 and 3.4 mmol/L (vildagliptin 10, 25 and 50 mg bid respectively, p<0.001 for all vs. PBO). Adverse events (AEs) were reported by 74% of patients receiving PBO and 62% in each of the vildagliptin dosage groups with a similar incidence of gastrointestinal AEs in all groups. Hypoglycemia occurred in 4.2, 0, 2.6 and 1.4% of patients with vildagliptin 10, 25, 50 mg bid and PBO respectively. This study indicates that treatment with vildagliptin reduces HbA?1c in a dose-dependent manner and is an effective and well-tolerated approach to achieving glycemic control in Japanese patients with T2DM.
Vildagliptin Is as Effective as Rosiglitazone in Lowering HbA1c but without Weight Gain in Drug-Naive Patients with Type 2 Diabetes (T2DM)
Year: 2006 Abstract Number: 557-P Authors: JULIO ROSENSTOCK, MICHELLE A. BARON, ANJA SCHWEIZER, DAVID MILLS, SYLVIE DEJAGE R. Institutions: Dallas, TX; E Hanover, NJ; Basel, Switzerland.
Results: Vildagliptin is a DPP-4 inhibitor that improves islet function by increasing ?- and ?-cell responsiveness to glucose. We report the findings of a 24-wk, multicenter, randomized, double-blind study comparing efficacy and tolerability of vildagliptin (50 mg bid, n=459) vs rosiglitazone (Rosi, 8 mg qd, n=238). The two groups were well-balanced at baseline (BL). Mean age=54.4yrs, BMI=32.4 kg/m?2, diabetes duration=2.4yrs and HbA?1c=8.7%. Figure 1 depicts mean HbA?1c during 24-wk treatment. The adjusted mean change (AM?) in HbA?1c from BL to endpoint in patients (pts) receiving vildagliptin 50 mg bid was -1.1±0.1% and non-inferiority to Rosi 8 mg qd was established. [figure1] In pts with BL HbA?1c >9.0%, the ?HbA?1c was -1.8±0.1% in the vildagliptin group (n=166) and -1.9±0.2% in the Rosi group (n=88). Relative to Rosi, vildagliptin improved lipid profiles, producing decreases in TG (-9%, P=0.010), total (-14%, P<0.001), LDL (-16%, P<0.001), non-HDL (-16%, P<0.001) and VLDL-cholesterol (-8%, P=0.007), but a smaller increase in HDL-cholesterol (-5%, P=0.003). Vildagliptin did not affect body weight (BW, AM?= -0.3±0.2 kg) but BW increased in Rosi-treated pts (AM?= 1.6±0.3 kg, P<0.001 vs vildagliptin). The overall incidence of side effects was 61.4% in pts receiving vildagliptin vs 64.0% with Rosi and the incidence of edema was greater with Rosi (4.9%) vs vildagliptin (2.5%). One patient in each group experienced one mild hypoglycemic episode. In conclusion, vildagliptin 50 mg bid is efficacious and well-tolerated in drug-naive pts with T2DM demonstrating similar glycemic reductions relative to Rosi 8 mg qd but without weight gain and with an improved lipid profile.
Four-Week Treatment with Vildagliptin Increases Fasting Plasma Levels of Intact Incretin Hormones Both in Patients with Type 1 (T1DM) and Type 2 (T2DM) Diabetes
Year: 2006 Abstract Number: 1481-P Authors: DAVID E. KELLEY, BETH E. DUNNING, MONICA LIGUEROS-SAYLAN, JENS J. HOLST, CAROLYN F. DEACON, JAMES E. FOLEY.
Institutions: Pittsburgh, PA; Princeton, NJ; E Hanover, NJ; Copenhagen, Denmark.
Results: Vildagliptin increases post-meal plasma levels of intact (active) GLP-1 and GIP in patients (pts) with T2DM by inhibiting DPP-4, the enzyme that degrades and inactivates these incretin hormones. Because incretin hormone release is dependent on nutrient ingestion, it might be expected that DPP-4 inhibition would have little influence on GLP-1 or GIP in the fasting state. Here we examine the effects of 4-wk Rx with vildagliptin (100 mg bid) on fasting plasma levels of intact GLP-1 and GIP in 11 insulin-pump treated pts with T1DM (mean age=39 y, BMI=24 kg/m?2) and in 9 drug-naive pts with T2DM (mean age=45 y, BMI=32 kg/m?2). Table 1 summarizes fasting (pre-dose) levels of intact GLP-1 and GIP at baseline (BL) and after 4-wk Rx. [table1] Both at BL and after 4-wk Rx, fasting plasma levels of intact GIP were significantly higher in obese pts with T2DM than in lean pts with T1DM. Prior to treatment (BL) fasting levels of intact GLP-1 were undetectable (<2 pM) in 5 of 9 pts with T2DM and in 9 of 11 pts with T1DM. Four-wk treatment with vildagliptin significantly increased fasting levels of active GLP-1 (by ~9 pM in T1DM and ~14 pM in T2DM) and GIP (>2-fold in both T1DM and T2DM). The magnitude of change (delta) in GLP-1 and GIP associated with 4-wk vildagliptin administration did not differ significantly between T1DM and T2DM. We conclude that 4-wk treatment with vildagliptin increases fasting plasma levels of intact (active) GLP-1 and GIP in the absence of an acute (meal) stimulus for incretin hormone release in pts with T1DM or with T2DM.
The Influence of Hepatic Impairment on the Pharmacokinetics of Vildagliptin
Year: 2006 Abstract Number: 2024-PO Authors: YAN-LING L. HE, RON SABO, YIBIN WANG, MONICA LIGUEROS-SAYLAN, WILLIAM P. DOLE. Institutions: Cambridge, MA; East Hanover, NJ.
Results:
Objectives: Vildagliptin is a potent DPP-4 inhibitor in clinical development for the treatment of type 2 diabetes (T2D). The primary metabolic pathway is hydrolysis and liver is one of the major organs that contributes to this process. The objective of this study was to investigate the effects of hepatic impairment (HI) on the PK of vildagliptin. Methods: This was an open label study in patients with mild (n=6), moderate (n=6) and severe (n=4) HI and subjects with normal hepatic function (n=6). Each subject received a single oral dose (100 mg) and blood samples were collected for measuring the concentrations of vildagliptin and its major metabolite (LAY151, pharmacologically inactive) with LC-MS/MS. Results: The PK parameters of vildagliptin and LAY151 are summarized in the table below. The exposure (AUC) to vildagliptin was increased by 30% in subjects with severe HI compared to the healthy subjects. There was considerable variability in the values for Cmax and AUC among all groups and there was no correlation between the exposure and the severity of HI. The elimination t?1/2 of vildagliptin was unaffected by HI. Exposure to the inactive metabolite (LAY151) increased in subjects with HI and correlated with the severity of HI. Vildagliptin 100 mg single dose was safe and well tolerated in subjects with HI. Conclusions: Exposure to vildagliptin is slightly increased in subjects with severe HI, however, this is not of clinically relevant. Therefore, no dose adjustment is necessary in subjects with HI based PK.[table1]
Tables:
|
PK
parameters of vildagliptin and LAY151
|
||||||
|
|
|
Vildagliptin
|
|
|
LAY151
|
|
|
|
Cmax (ng/mL)
|
AUC0-inf (hxng/mL)
|
t½ (h)
|
Cmax (ng/mL)
|
AUC0-inf (hxng/mL)
|
t½ (h)
|
|
Healthy
|
675 ± 263
|
2580 ± 425
|
2.01 ± 0.50
|
330 ± 99
|
5641 ± 1884
|
7.93 ± 0.67
|
|
Mild
|
497 ± 229
|
2101 ± 512
|
4.92 ± 4.86
|
406 ± 108
|
7252 ± 1782
|
8.36 ± 1.57
|
|
Moderate
|
512 ± 166
|
2437 ± 742
|
3.08 ± 1.59
|
483 ± 165
|
8396 ± 3556
|
7.19 ± 0.76
|
|
Severe
|
632 ± 247
|
3354 ± 1462
|
2.40 ± 0.25
|
549 ± 217
|
11545 ± 5964
|
9.32 ± 1.51
|
Year: 2006 Abstract Number: 5-LB Authors: KOICHIRO AZUMA, ZOFIA RADIKOVA, JULIET M. MANCINO, FREDERICO G. S. TOLEDO, ERNESTINE THOMAS, NOAH D. LUBOWSKY, CAROL A. KELLEY, CYROUS O. KANGANI, DENISE SERRA, YANLING HE, MONICA LIGUEROS-SAYLAN, JAMES E. FOLEY, DAVID E. KELLEY Institutions: Pittsburgh, PA; East Hanover, NJ
Results: Mechanisms by which incretin hormones improve glucose homeostasis in type 2 DM may involve extra-pancreatic effects as well as those upon islet function. To address this question, a two-step insulin infusion clamp study was performed to assess effects of vildagliptin (V), a dipeptidyl dipeptidase 4 (DPP4) inhibitor, versus placebo (P) on IR in type 2 DM. V was given at 50 mg bid for 6 weeks in a randomized order, double-blinded, placebo-controlled within-subject cross-over study design. 16 type 2 DM (F7M9, Age 55±7, BMI 31±4, HbA1c 7.1±0.7) participated, 11 subjects were also on metformin; other anti-diabetic medications were excluded. The two-step clamp consisted of 3hr at 20 mU/min-m?2 (LO) with isoglycemia followed by 2hr at 80 mU/min-m?2 (HI) with euglycemia. [6, 6 ?2H?2] glucose and [U-?13C] palmitate were infused to assess glucose and palmitate flux. On the day preceding clamps, a standardized mixed meal study was performed. Fasting glucose (123±17 vs 143±24 mg/dl; p<0.01), and insulin (7.8±4.9 vs 8.9±5.8 uU/ml; p<0.05) were lower with V than P, as was fasting palmitate flux (120±31 vs 145±43 umol/min; p<0.01). Fasting endogenous glucose production (EGP) was similar (1.76±0.19 vs 1.80±0.21 mg/min-kg; V vs P), but systemic glucose clearance (MCR) was higher with V during fasting (1.45±0.26 vs 1.30±0.26 ml/min-kg; p<0.01), and LO (1.83±0.61 vs 1.58±0.51; p<0.01), and during HI, glucose disposal was greater with V (6.05±1.94 vs 5.35±2.03 mg/min-kg; p<0.05). Plasma insulin was matched in V vs P at LO and HI. During the meal study, calculated insulin sensitivity was higher with V vs P (3hr-OGIS: 345±14 vs 294±10 ml/min-m?2; p<0.001). In summary, vildagliptin improves IR by ~15%, indicating that DPP4 inhibition contributes to improved glucose homeostasis in type 2 DM through extra-pancreatic effects.
